💎
Block C · Lectures 7–9
Aromatics & Heterocycles
Benzene aromaticity, Hückel's rule, electrophilic aromatic substitution with directing effects, and the heterocyclic rings found in DNA bases and blockbuster drugs like Lipitor and Gleevec.
3
Lectures
30
MCQs
9
Structured Qs
4
SVG Diagrams
Lectures
Block Progress
Lec 7: Aromatic Hydrocarbons I
Lec 8: Aromatic Hydrocarbons II
Lec 9: Heterocyclic Compounds in Biology
Lecture 7
Aromatic Hydrocarbons I
Benzene structure, aromaticity, Hückel's rule, and electrophilic aromatic substitution
Learning Objectives
- 1.Recall the structure of benzene
- 2.Define 'resonance'
- 3.Recall the bonding in benzene
- 4.State Hückel's rule for aromaticity
- 5.Identify benzenoid and non-benzenoid aromatic compounds
- 6.Mechanistically describe 'electrophilic substitution reactions'
- 7.Recall products and mechanism of halogenation, Friedel-Crafts alkylation, Friedel-Crafts acylation and nitration of aromatic hydrocarbons
The Structure of Benzene
- Benzene (C₆H₆) was first isolated by Michael Faraday in 1825 (1791–1867). It is a colourless, toxic, carcinogenic liquid with a boiling point of 80°C.
- Structure: Six carbon atoms joined in a ring, one hydrogen on each carbon. All C–C bonds are equal length at 139 pm (intermediate between single 154 pm and double 134 pm). Bond angle = 120°.
- F. August Kekulé (1829–1896) proposed the ring structure. He dreamed of a snake seizing its own tail — the Ouroboros symbol — inspiring the cyclic structure. However, his cyclohexatriene model predicted alternating bond lengths, contradicting experiment.
- Resonance: Benzene is a resonance hybrid of two Kekulé structures. Individual resonance forms are imaginary, not real — the real structure is a composite. Important: Do NOT confuse resonance (↔) with equilibria (⇌). Benzene is NOT two structures in equilibrium — it is a single structure that is a resonance hybrid.
- Orbital model: Each carbon is sp² hybridised, forming three σ bonds. The remaining 2pz orbital on each carbon overlaps sideways with its two neighbours, creating a continuous π electron cloud containing 6π electrons (an 'aromatic sextet') delocalised over all six carbons.
- Aromatic stability — Heats of hydrogenation: Cyclohexene → cyclohexane: −118 kJ/mol. 1,3-Cyclohexadiene → cyclohexane: −230 kJ/mol. Expected for benzene (3 double bonds): −356 kJ/mol. Actual measured value: −206 kJ/mol. The difference of 150 kJ/mol = extra stability due to aromaticity.
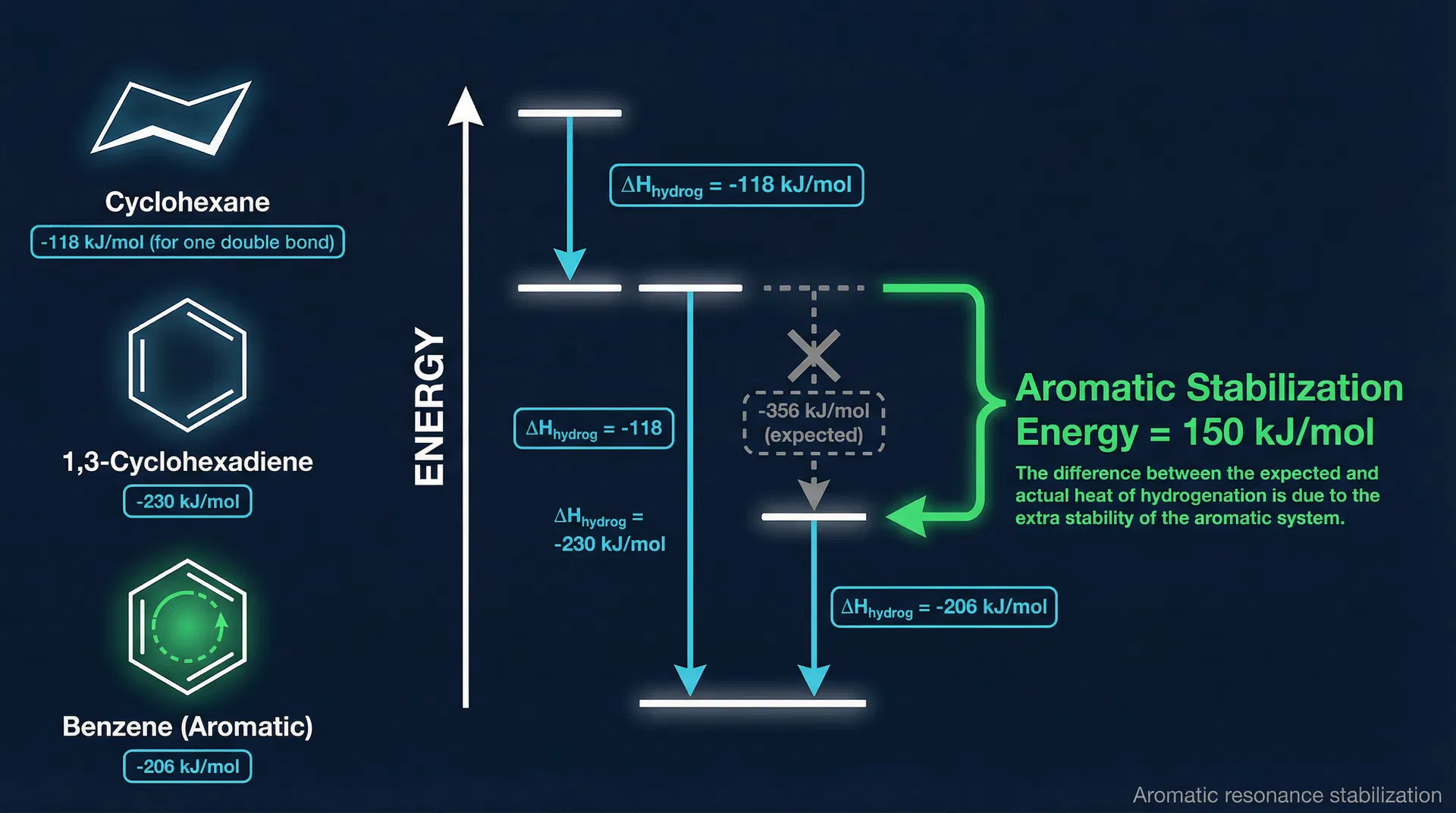
Key Points
- ◆All 6 C–C bonds in benzene are equal length (139 pm) — evidence for delocalisation
- ◆Each carbon is sp² hybridised — trigonal planar, bond angle 120°
- ◆6π electrons delocalised over the ring (aromatic sextet)
- ◆Resonance hybrid: use ↔ (resonance arrow), NOT ⇌ (equilibrium arrow)
- ◆Delocalisation energy = 150 kJ/mol (from heats of hydrogenation data)
- ◆Faraday (1825) isolated benzene; Kekulé (1865) proposed ring structure (Ouroboros dream)
Hückel's Rule and Aromatic Compounds
- Hückel's rule (1931): To be aromatic, a compound must have: (1) a planar ring, (2) each atom in the ring has a pz orbital, and (3) (4n + 2)π electrons where n is an integer.
- Examples: Benzene (n = 1 → 6π ✓), Naphthalene (n = 2 → 10π ✓), Anthracene (n = 3 → 14π ✓)
- Anti-aromatic: 4n π electrons — destabilised. Cyclobutadiene (4π) is extremely reactive.
- Named aromatic compounds: Toluene (methylbenzene, CH₃ group), Aniline (aminobenzene, NH₂ group), Phenol (hydroxybenzene, OH group)
- Naming di-substituted benzenes: ortho (o) = 1,2 positions; meta (m) = 1,3 positions; para (p) = 1,4 positions
- Examples: ortho-dibromobenzene = 1,2-dibromobenzene; meta-nitrophenol = 3-nitrophenol; para-nitrophenol = 4-nitrophenol
- Drug derivatives: Many drugs are benzene derivatives and are synthesised from benzene or substituted benzenes.
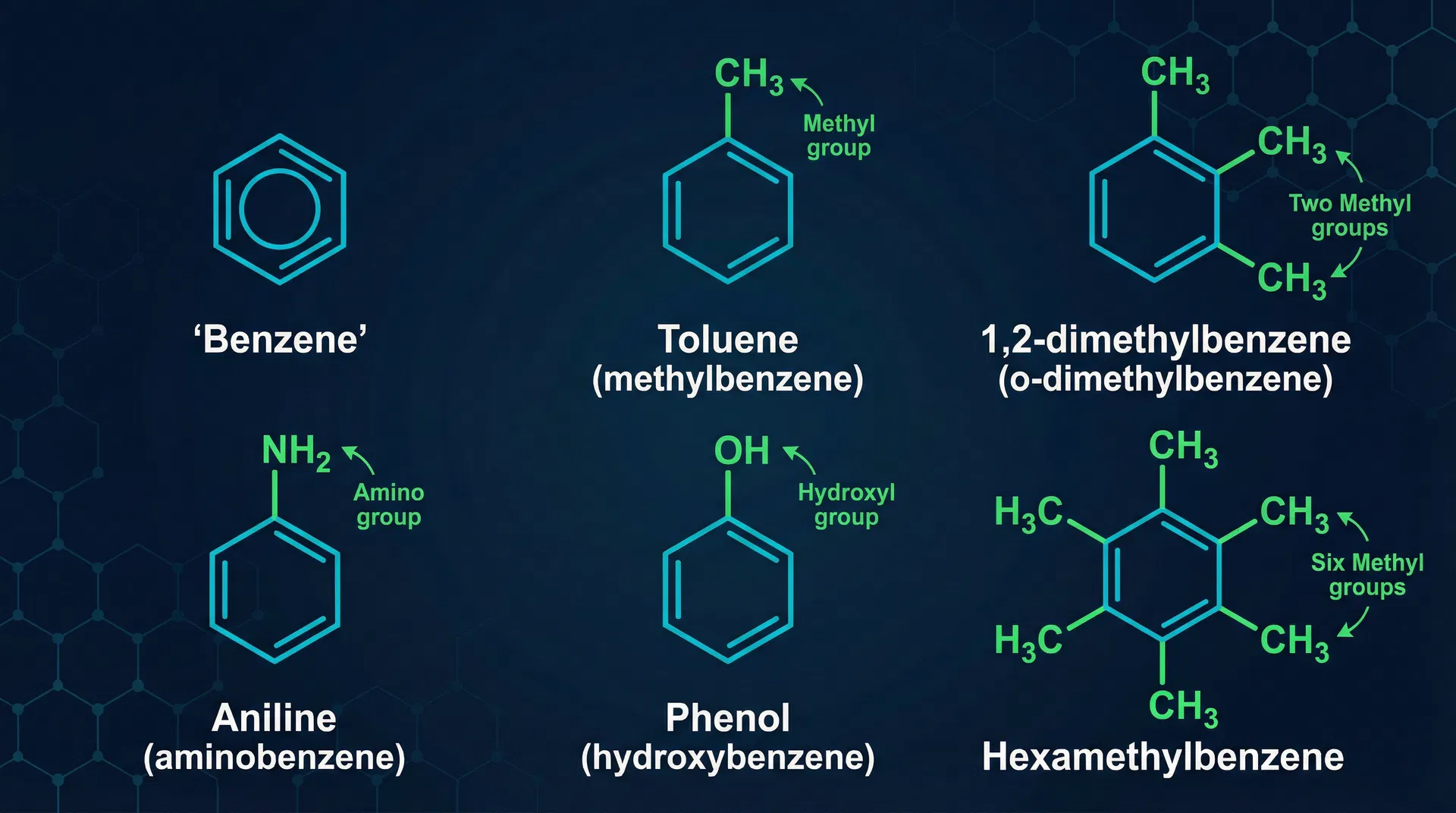
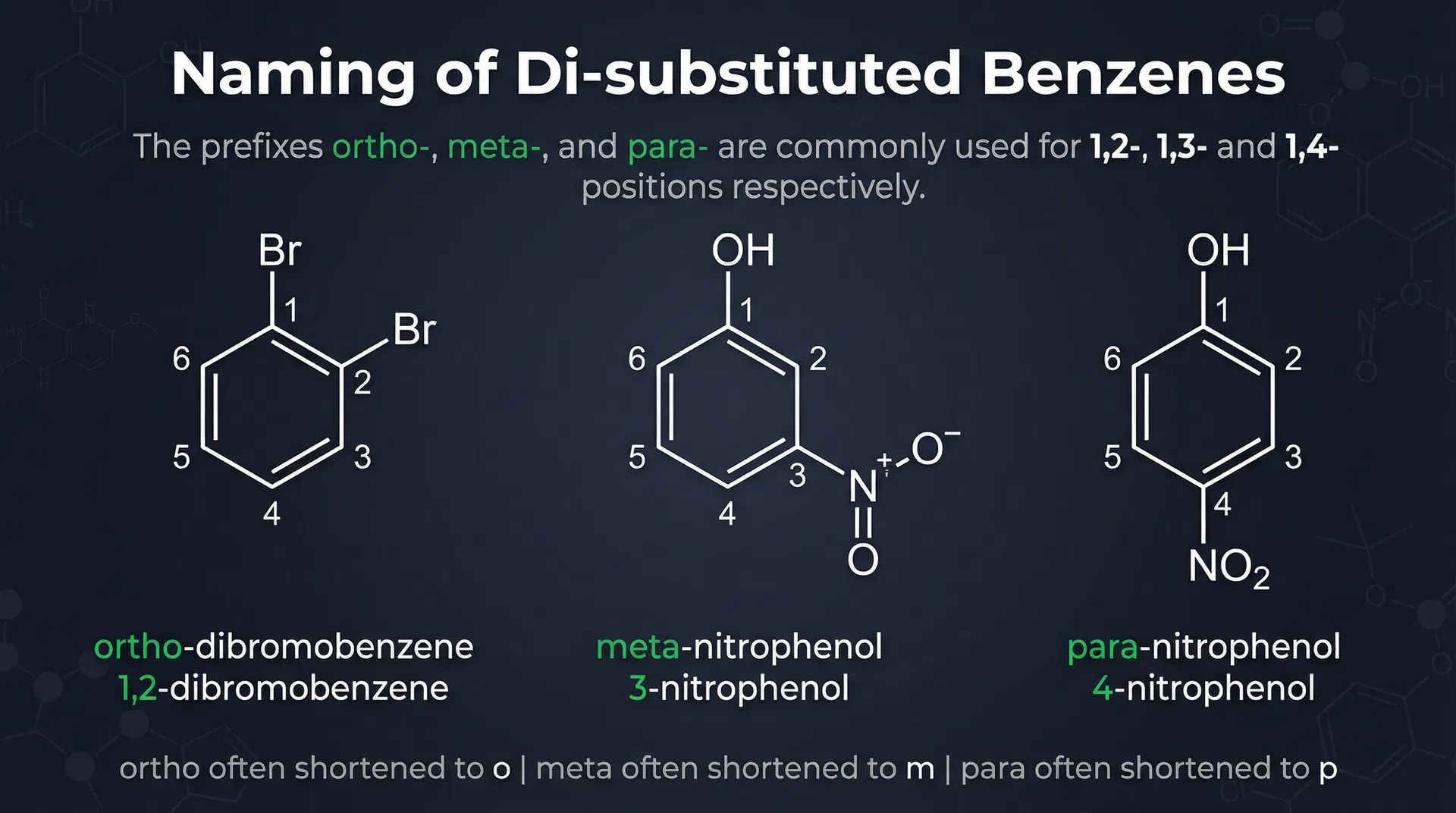
Key Points
- ◆Aromatic: planar ring + p_z orbitals on each atom + (4n+2)π electrons (n = integer)
- ◆Benzene: 6π (n=1) | Naphthalene: 10π (n=2) | Anthracene: 14π (n=3)
- ◆Anti-aromatic: 4n π electrons — cyclobutadiene (4π) is extremely reactive
- ◆Naming: ortho (1,2), meta (1,3), para (1,4)
- ◆Named compounds: toluene, aniline, phenol
Electrophilic Aromatic Substitution (EAS)
- Curly arrows: Used to describe reactions, showing how chemical bonds are made and broken. A curly arrow represents the actual movement of a pair of electrons from source to destination. A covalent bond is made of a shared pair of electrons.
- Benzene reactivity: Despite its stability, benzene does react — but it strongly prefers substitution over addition. Substitution reactions preserve the aromatic sextet (6π electrons). These are electrophilic substitution because electrophiles react with the electron-rich aromatic ring.
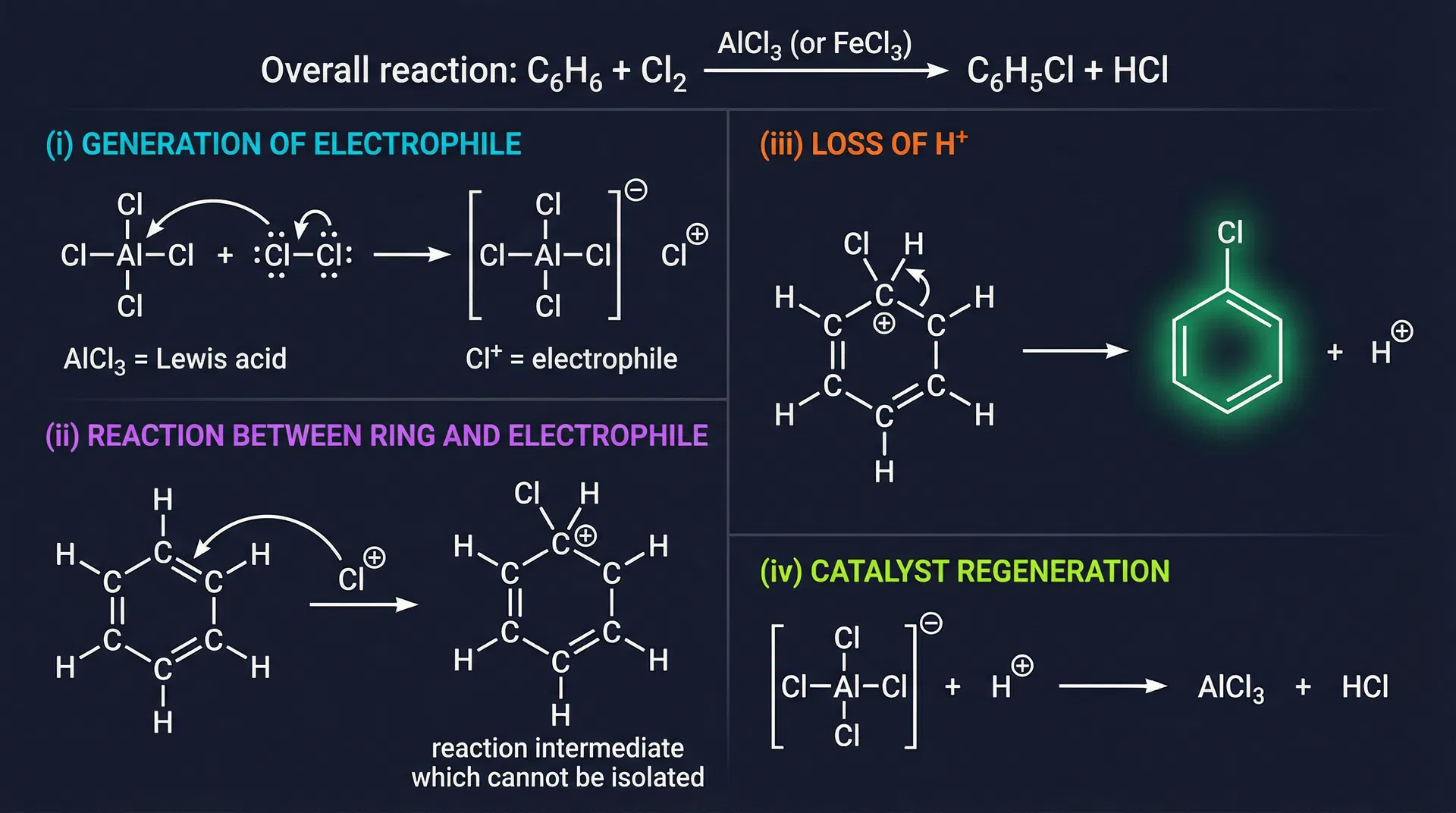
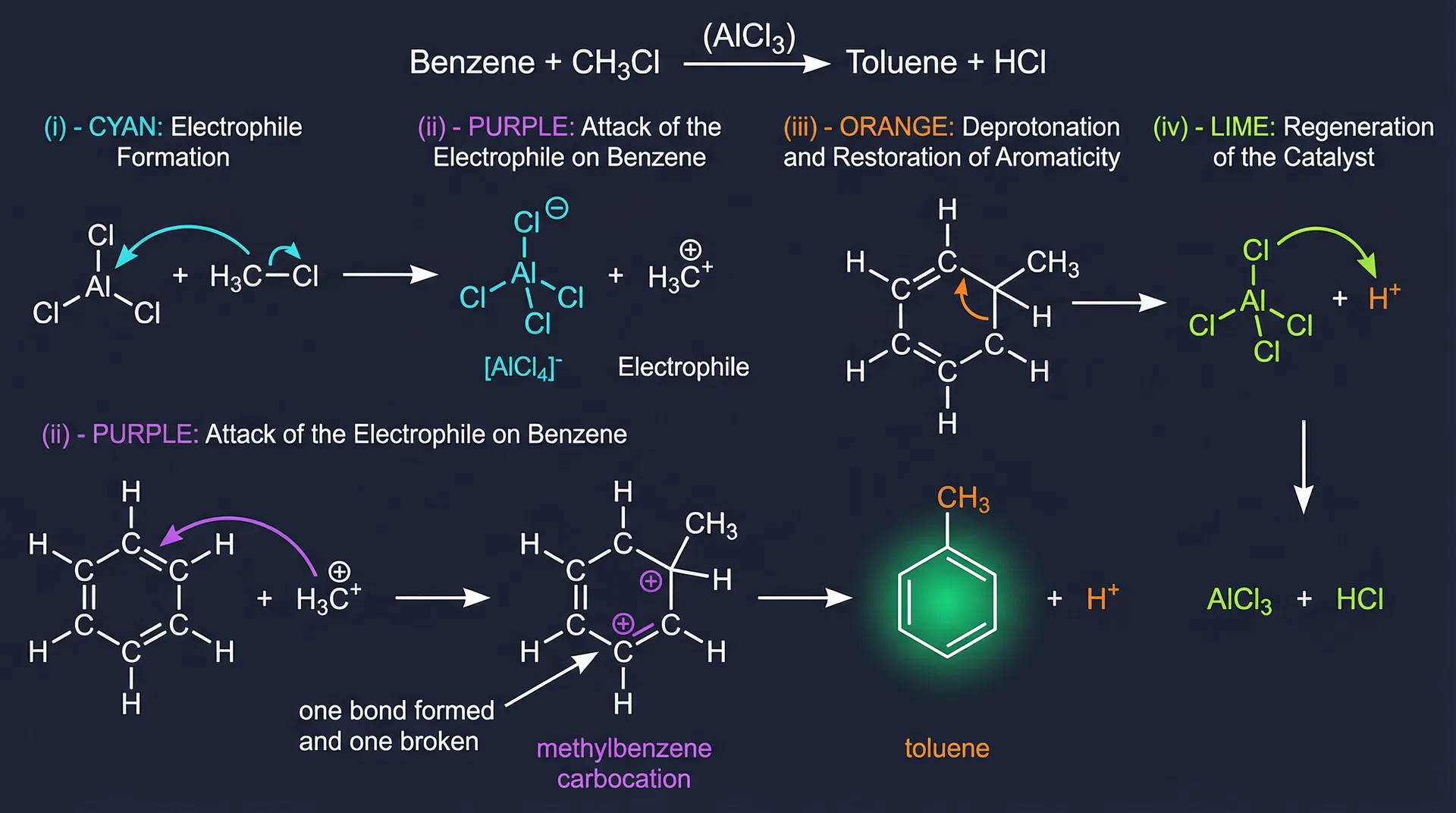
- Step (i) — Generation of electrophile: A Lewis acid catalyst (e.g., AlCl₃) activates the reagent. AlCl₃ acts as a Lewis acid (electron pair acceptor) — it has only 6 electrons around the central atom. It abstracts Cl⁻ from Cl₂, generating the electrophile Cl⁺. Cl⁻ acts as a Lewis base (electron pair donor). Remember: BF₃ and all Group III trihalides (e.g., AlCl₃) are Lewis acids.
- Step (ii) — Electrophile attacks ring: The electrophile (E⁺) attacks the π electron cloud of benzene, forming a carbocation intermediate called the benzonium ion (arenium ion). This intermediate is NOT aromatic — one carbon is now sp³.
- Step (iii) — Loss of H⁺: A base removes a proton from the sp³ carbon, restoring aromaticity. The driving force is the regeneration of aromaticity.
- Step (iv) — Catalyst regeneration: The conjugate base of the catalyst (e.g., [AlCl₄]⁻) reacts with H⁺ to regenerate the catalyst (AlCl₃ + HCl).
Key Points
- ◆EAS mechanism: 4 steps — (i) generate E⁺, (ii) E⁺ attacks ring → benzonium ion, (iii) loss of H⁺, (iv) catalyst regenerated
- ◆Benzonium ion (arenium ion) is NOT aromatic — one carbon is sp³
- ◆Driving force for step (iii) = regeneration of aromaticity
- ◆Curly arrows show movement of electron pairs (source → destination)
- ◆AlCl₃ = Lewis acid (electron pair acceptor, 6 electrons around Al)
Common EAS Reactions
- Halogenation (chlorination): Benzene + Cl₂ → (AlCl₃ or FeCl₃ catalyst) → chlorobenzene + HCl. The 4-step mechanism: (i) AlCl₃ + Cl₂ → [AlCl₄]⁻ + Cl⁺; (ii) Cl⁺ attacks ring → benzonium ion; (iii) loss of H⁺ → chlorobenzene; (iv) [AlCl₄]⁻ + H⁺ → AlCl₃ + HCl.
- Bromination: Br₂ reacts in presence of AlBr₃ catalyst. F₂ reacts violently without catalyst. No light is needed (not a free radical mechanism).
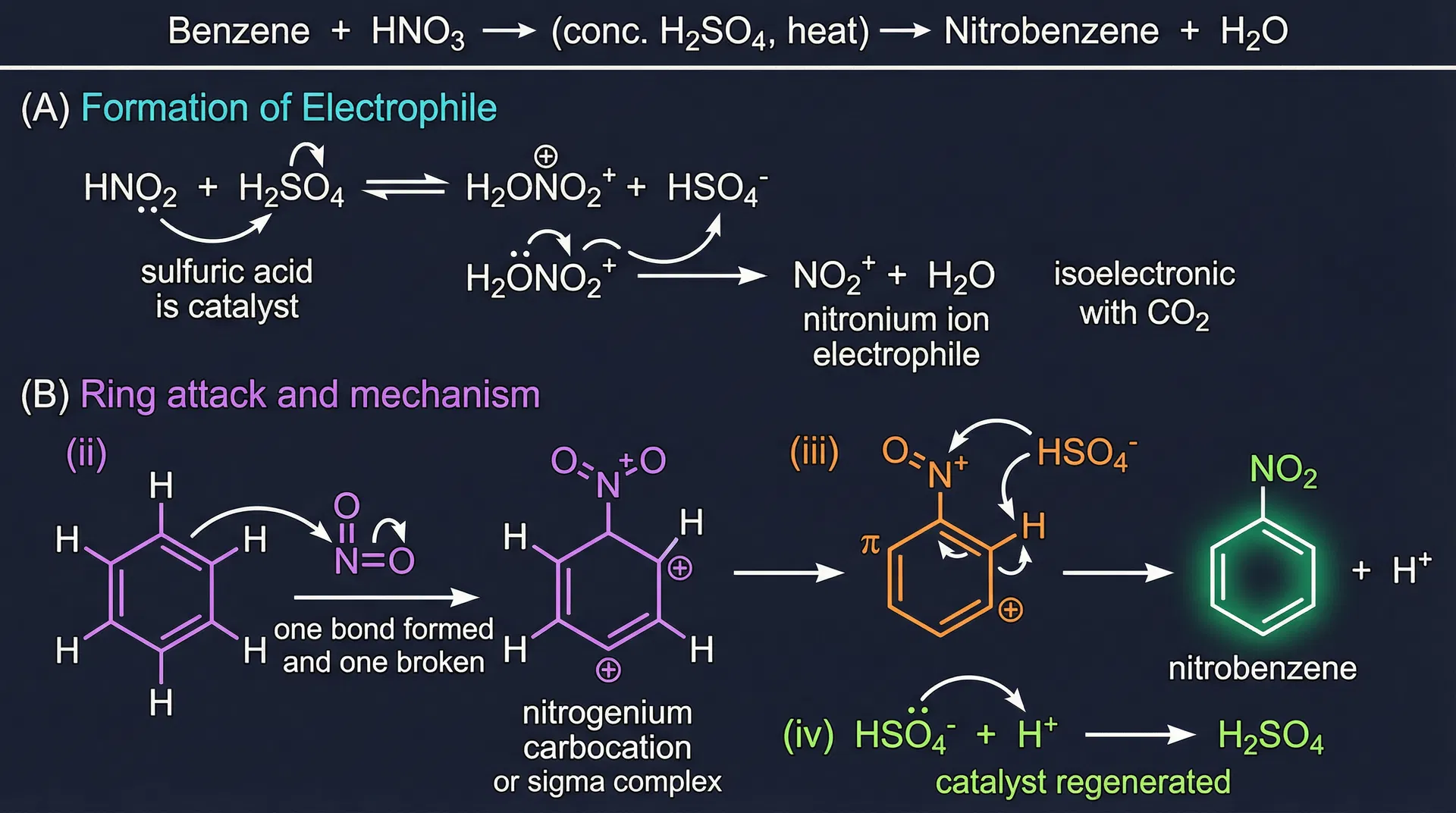
- Nitration: Benzene + HNO₃ → (conc. H₂SO₄, heat) → nitrobenzene + H₂O. Formation of electrophile: H₂SO₄ protonates HNO₃ → H₂ONO₂⁺ + HSO₄⁻, then H₂ONO₂⁺ → NO₂⁺ (nitronium ion) + H₂O. NO₂⁺ is isoelectronic with CO₂. The mechanism follows the same 4 steps: (ii) NO₂⁺ attacks ring; (iii) loss of H⁺; (iv) HSO₄⁻ + H⁺ → H₂SO₄ (catalyst regenerated).
- Friedel-Crafts Alkylation (Charles Friedel, 1832–1899, Sorbonne; James Crafts, 1839–1917, MIT): Benzene + CH₃Cl → (AlCl₃) → toluene + HCl. These are important carbon–carbon bond forming reactions. Mechanism: same 4 steps — AlCl₃ + CH₃Cl → [AlCl₄]⁻ + CH₃⁺. Note: Reaction does NOT work with aryl halides (e.g., chlorobenzene) or vinylic halides (e.g., chloroethene) where Cl is attached directly to an unsaturated carbon, as these carbocations are too difficult to form.
- Friedel-Crafts Acylation: Benzene + RCOCl + AlCl₃ → aryl ketone. The acylium ion (RCO⁺) is the electrophile. Unlike alkylation, only one acylation occurs because the carbonyl group deactivates the ring.
Key Points
- ◆Chlorination: Cl₂/AlCl₃ → Cl⁺ electrophile → chlorobenzene (4-step mechanism)
- ◆Bromination: Br₂/AlBr₃; F₂ reacts violently without catalyst
- ◆Nitration: HNO₃/H₂SO₄ → NO₂⁺ (nitronium ion, isoelectronic with CO₂) → nitrobenzene
- ◆Friedel-Crafts alkylation: RCl/AlCl₃ → R⁺ → C–C bond formation (Friedel & Crafts)
- ◆Friedel-Crafts does NOT work with aryl or vinylic halides