Topic 1
States of Matter
4 subtopics · Cambridge IGCSE Chemistry 0620
Key Idea
All matter is made of tiny particles in constant motion. Their arrangement and movement explains the properties of solids, liquids, and gases.
Explanation
SOLID: Fixed shape and volume. Particles very close together, vibrate in fixed positions. Very strong forces. Cannot flow or be compressed.
LIQUID: Fixed volume, no fixed shape. Particles close but can move past each other. Weaker forces. Can flow, almost incompressible.
GAS: No fixed shape or volume. Particles very far apart. Move rapidly in all directions. Very weak forces. Easily compressed.
Particle arrangement comparison — solid lattice vs liquid flow vs gas dispersion
💡 Analogy
Think of particles in a solid as people standing tightly packed in a crowded lift — they can only vibrate. In a liquid they are like people in a busy corridor — they can move past each other. In a gas they are like people in a large open field — they move freely in all directions.
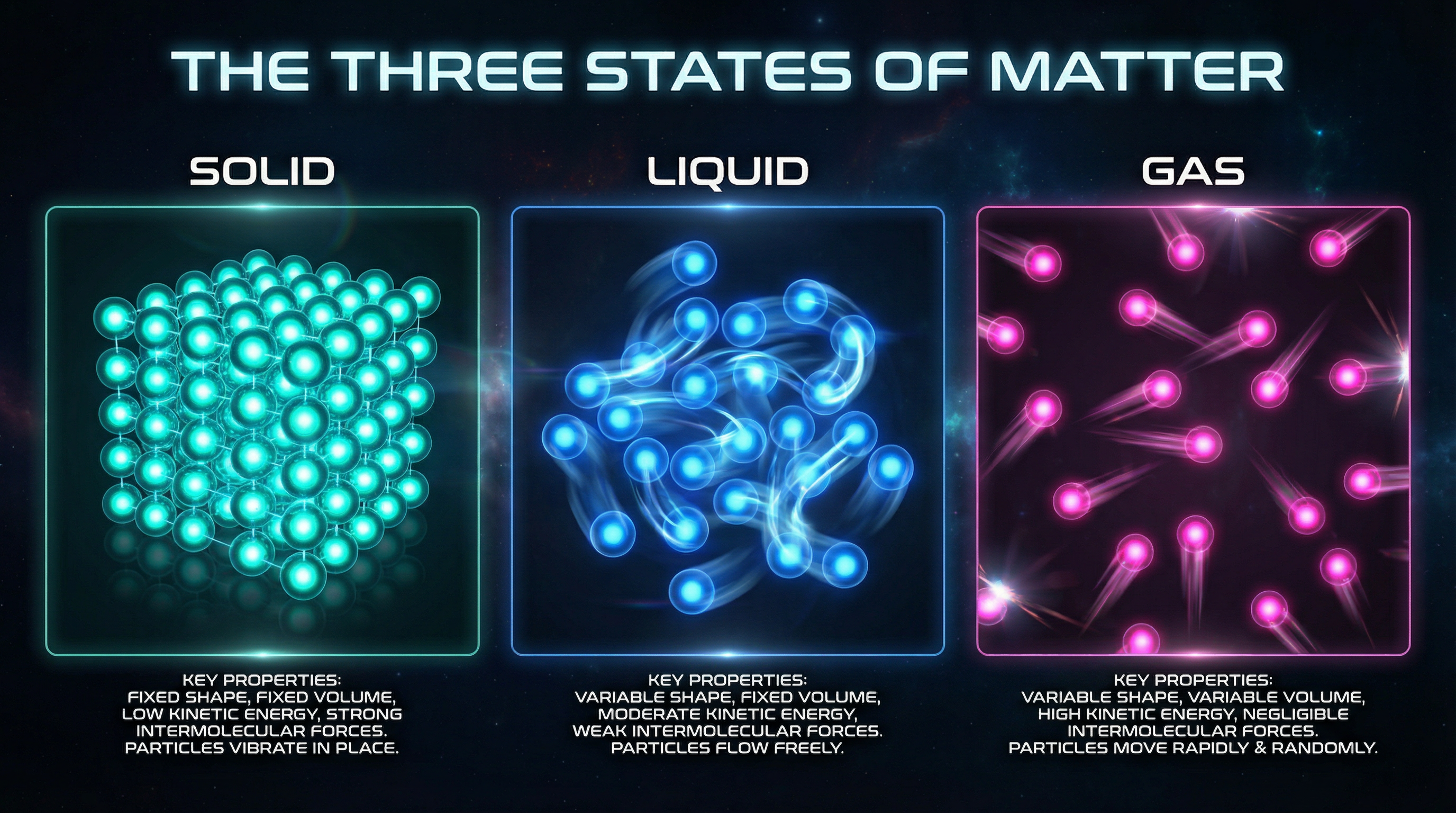
The Three States of Matter — particle arrangement, motion, and key properties visualised
Practice Questions
Which of the following best describes the arrangement and movement of particles in a liquid?
Key Idea
When a substance is heated or cooled it can change state. During a change of state, temperature stays constant — energy is used to break or form forces between particles, NOT to raise temperature.
Explanation
Changes of state:
- SOLID to LIQUID: Melting. Energy absorbed, forces weaken.
- LIQUID to GAS: Boiling or Evaporation. Energy absorbed, forces overcome.
- SOLID to GAS: Sublimation (e.g. iodine, dry ice).
- LIQUID to SOLID: Freezing. Energy released.
- GAS to LIQUID: Condensation. Energy released.
BOILING vs EVAPORATION:
Boiling occurs at a specific temperature (boiling point) throughout the whole liquid.
Evaporation occurs at any temperature, only at the surface. Evaporation causes cooling because the most energetic particles escape.
HEATING CURVE:
Temperature rises in a straight line, then stays flat during melting and boiling. The flat sections show where energy is used to break forces between particles. A pure substance has a sharp, single melting point; an impure substance melts over a range.
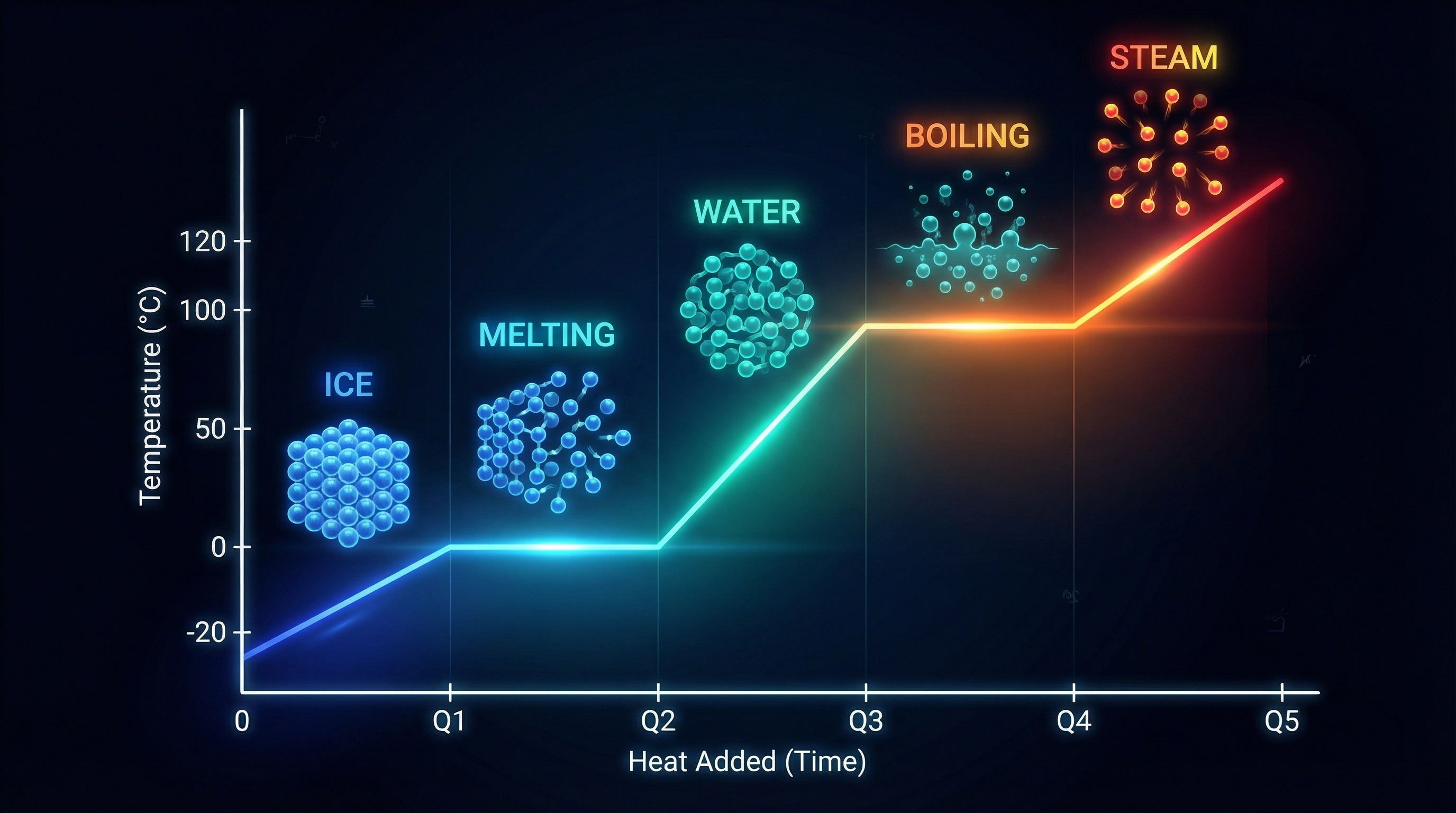
Heating Curve for Water — temperature vs heat added, showing flat plateaus at melting (0°C) and boiling (100°C) points with particle illustrations
Worked Examples
Practice Questions
A student heats a pure solid substance and records the temperature every minute. Describe what happens to the temperature during melting, and explain this in terms of particles. [3 marks]
What happens to the temperature of a pure substance during melting?
Explain, in terms of particles, why the boiling point of water is higher than that of ethanol.
A student heats ice from -10 degrees C to 110 degrees C. Describe what happens at 0 degrees C and at 100 degrees C.
Sketch a heating curve for a pure substance from solid to gas and explain the shape at each stage.
Key Idea
Diffusion is the net movement of particles from a region of higher concentration to a region of lower concentration, as a result of the random motion of particles.
Explanation
Diffusion continues until particles are evenly spread (equilibrium).
Factors that INCREASE diffusion rate:
- Higher temperature: particles have more kinetic energy, move faster.
- Lower molecular mass: lighter particles move faster.
- Steeper concentration gradient.
- Gas vs liquid: diffusion is much faster in gases.
Factors that DECREASE diffusion rate:
- Lower temperature.
- Higher molecular mass.
- Smaller concentration gradient.
💡 Analogy
When you spray perfume in one corner of a room, the scent molecules move randomly in all directions. Over time, the smell spreads throughout the entire room — from high concentration to low concentration. This is diffusion!
Worked Examples
Practice Questions
Two cotton wool plugs are placed at opposite ends of a glass tube. One is soaked in ammonia (Mr = 17) and the other in hydrochloric acid (Mr = 36.5). A white ring of ammonium chloride forms inside the tube. Where does the ring form?
Which factor increases the rate of diffusion of a gas?
Explain, in terms of particles, why a drop of purple ink spreads throughout a beaker of still water. [2 marks]
Gas A has Mr = 4 and gas B has Mr = 64. Both are released from opposite ends of a tube at the same time. State which gas diffuses faster and explain why. [2 marks]
A student opens a bottle of perfume at one end of a room. Explain why a person at the other end of the room can smell the perfume after a short time, and why it takes longer on a cold day. [3 marks]
Key Idea
Gas pressure is caused by the constant collision of gas particles with the walls of their container. Boyle's Law: P x V = constant (at constant temperature).
Explanation
How pressure changes:
1. Increase temperature: particles move faster, more frequent and harder collisions with walls, pressure increases (at constant volume).
2. Decrease volume: particles are closer together, more frequent collisions with walls, pressure increases (at constant temperature). This is Boyle's Law: P x V = constant.
3. Add more gas particles: more particles, more collisions per second, pressure increases.
4. Increase volume: particles travel further between collisions, fewer collisions per second, pressure decreases.
Worked Examples
Practice Questions
A sealed syringe contains 250 cm3 of gas at 120 kPa. The piston is pushed in until the volume is 100 cm3. Temperature remains constant.
a) Calculate the new pressure. Show your working. [2]
b) Explain, in terms of particles, why the pressure increased. [2]
A gas exerts pressure on the walls of its container. Which explanation is correct?
Explain why increasing the temperature of a gas in a sealed container increases the pressure.
A fixed amount of gas has a volume of 500 cm3 at 100 kPa. Calculate its volume at 200 kPa, assuming temperature is constant.
Explain, using kinetic particle theory, why a gas is much less dense than a liquid of the same substance.