Chapter 3 · Comprehensive Revision
Bonds and Forms of Molecules
4 lessons · 177 total questions
Chapter Overview & Mind Map
This chapter explores the fundamental principles of atomic structure, bonding types, and molecular geometry. Master these concepts to understand how atoms connect and why molecules behave the way they do.
🗺️ Comprehensive Mind Map
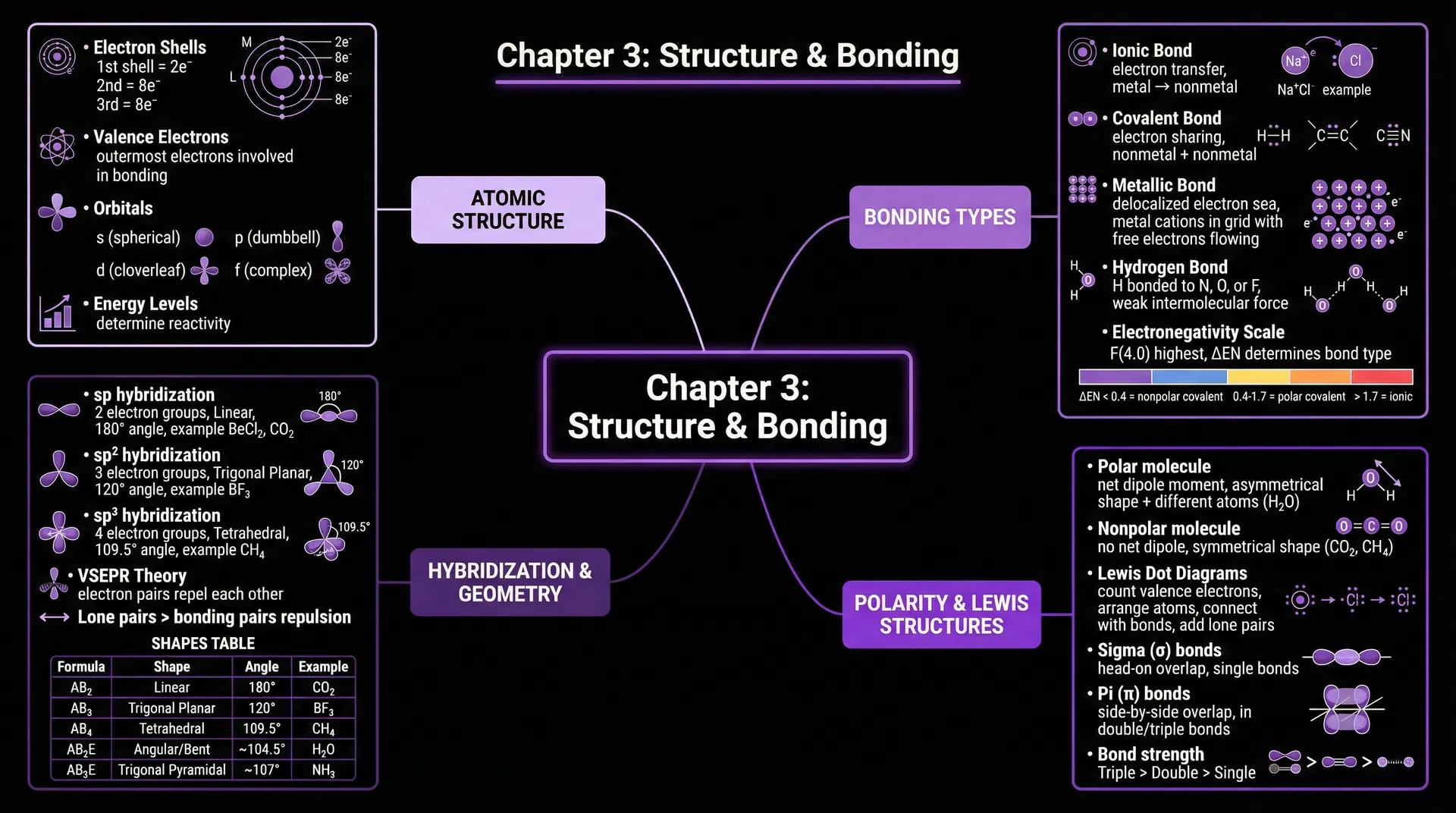
🔬 Atomic Structure
- • Electrons in shells (1st: 2, 2nd: 8)
- • Valence electrons (bonding electrons)
- • Orbitals: s, p, d, f shapes
- • Energy levels determine reactivity
⚛️ Bonding Types
- • Ionic: electron transfer
- • Covalent: electron sharing
- • Metallic: delocalized electrons
- • Hydrogen: weak intermolecular
🔷 Molecular Geometry
- • VSEPR theory (electron repulsion)
- • Hybridization: sp, sp², sp³
- • Molecular shapes & bond angles
- • Polarity & molecular properties
Lessons
4
in this chapter
Topics
7
key concepts
Questions
46
ready to practice
Ready to Test Yourself?
Take a timed practice test with instant feedback, score tracking, and a full answer review. Choose MCQ-only or the full test.